655. Anhalonine
Nomenclature
CAS number: 519-04-0
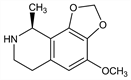
(9S)-6,7,8,9-Tetrahydro-4-methoxy-9-methyl-1,3-dioxolo[4,5-h]isoquinoline. C12H15NO3; mol wt 221.25.
C 65.14%, H 6.83%, N 6.33%, O 21.69%.
Description and references
From mescal buttons [Lophophora williamsii (Lemaire) Coult. (Anhalonium lewinii Henn.), Cactaceae] also in Ariocarpus, in Gymnocalycium gibbosum. Synthesis of dl-form and resolution: Spth, Kesztler, Ber. 68, 1663 (1935); Brossi et al., J. Am. Chem. Soc. 93, 6248 (1971). Configuration: Battersby, Edwards, J. Chem. Soc. 1960, 1214.
Properties
Rhombic needles from petr ether, mp 86°, bp0.02 140°. [α]D25 63.8° (methanol); 56.3° (chloroform). Very sol in alcohol, ether, chloroform, benzene, petr ether.Derivative
Hydrochloride.C12H15NO3.HCl; mol wt 257.71.
C 55.93%, H 6.26%, N 5.44%, O 18.62%, Cl 13.76%.