691. Anthrone
Nomenclature
CAS number: 90-44-8
9(10H)-Anthracenone; 9,10-dihydro-9-oxoanthracene; Carbothrone. C14H10O; mol wt 194.23.
C 86.57%, H 5.19%, O 8.24%.
Description and references
Prepn from anthraquinone by reduction with tin, glacial acetic and hydrochloric acid: Org. Synth. coll. vol. I, 60 (1941). By cyclization of o-benzylbenzoic acid with liquid HF: Fieser, Hershberg, J. Am. Chem. Soc. 61, 1278 (1939).
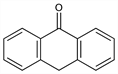
Properties
Orthorhombic needles from benzene + petr ether, mp 155°. Absorption spectrum: Martin, Ann. Combustibles Liq. 12, 967 (1937). Sol in most organic solvents without fluorescence. Any fluorescence present is due to anthranol. Tendency to change to anthraquinone. Equilibrium in abs alc: 89% anthrone; 11% anthranol.Derivative
Addition compound with 4 mols desoxycholic acid.C110H170O17; mol wt 1764.52.
C 74.87%, H 9.71%, O 15.41%.