736. Aplasmomycin
Nomenclature
Description and references
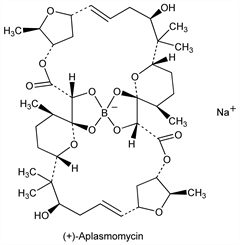
Antibiotic produced by Streptomyces griseus strain SS-20, obtained from shallow sea mud: Y. Okami et al., J. Antibiot. 29, 1019 (1976); eidem, JP Kokai 77 108901 (1977 to Microbiochem. Res. Found.), C.A. 88, 35843 (1978). It inhibits the growth of gram-positive bacteria in vitro and is active vs Plasmodium berghei in vivo. Aplasmomycin is a symmetrical dimer related to boromycin, q.v., the only other known natural product that contains boron. Structure, x-ray crystallography: H. Nakamura et al., J. Antibiot. 30, 714 (1977). Total synthesis of (+)-form: E. J. Corey et al., J. Am. Chem. Soc. 104, 6816, 6818 (1982); T. Nakata et al., Tetrahedron Lett. 27, 6341, 6345 (1986); J. D. White et al., J. Am. Chem. Soc. 108, 8105 (1986). Use as growth promotant in ruminants: D. H. Davies et al., EP 2893; eidem, US 4225593 (1979, 1980 both to ICI). Two minor components, aplasmomycins B and C, have also been isolated from the fermentation; both aplasmomycin and aplasmomycin B show ionophoric properties, mediating net K+ transport across a bulk phase: K. Sato et al., J. Antibiot. 31, 632 (1978). NMR analysis of aplasmomycin and deboroaplasmomycin: T. S. S. Chen et al., ibid. 33, 1316 (1980). Comparative anti-anaerobic activity of aplasmomycin: K. Watanabe et al., Antimicrob. Agents Chemother. 19, 519 (1981). Structural studies: T. J. Stout et al., Tetrahedron 47, 3511 (1991). Review of biosynthetic studies: H. G. Floss, C. Chang in Antibiotics IV, J. W. Corcoran, Ed. (Springer-Verlag, New York, 1981) pp 203-210.