746. Apomorphine
Nomenclature
Description and references
Dopamine (D1 and D2) receptor agonist. Synthetic opiate obtained by treating morphine with concd HCl: A. Matthiessen, C. R. A. Wright, Proc. R. Soc. London Ser. B 17, 455 (1869). Structure: R. Pschorr et al., Ber. 35, 4377 (1902). Configuration: H. Corrodi, E. Hardegger, Helv. Chim. Acta 38, 2038 (1955). Total synthesis of (±)-form: J. L. Neumeyer et al., J. Pharm. Sci. 59, 1850 (1970); of (+)- and ()-forms: V. J. Ram, J. L. Neumeyer, J. Org. Chem. 46, 2830 (1981). Toxicity data: J. Z. Ginos et al., J. Med. Chem. 18, 1194 (1975). Clinical evaluation in impotence: J. P. W. Heaton et al., Urology 45, 200 (1995). Historical review: J. L. Neumeyer et al., in Apomorphine and Other Dopaminomimetics vol. 1, G. L. Gessa, G. U. Corsini, Eds. (Raven, New York, 1981) p 1-17. Comprehensive description: F. J. Muhtadi, M. S. Hifnawy, Anal. Profiles Drug Subs. 20, 121-166 (1991). Review of pharmacology and clinical efficacy in Parkinson's disease: D. Muguet et al., Biomed. Pharmacother. 49, 197-209 (1995); in erectile dysfunction: F. Giuliano, J. Allard, Int. J. Impot. Res. 14, Suppl. 1, S53-S56 (2002).
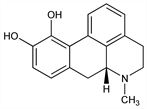
Properties
Hexagonal plates from chloroform and petr ether, dec 195°; subl in high vacuum. Oxidizes rapidly in air and becomes green. Sol in alcohol, acetone, chloroform. Slightly sol in water, benzene, ether, petr ether. Solns darken rapidly. pKb 7.0; pKa 8.92. uv max (98% alc): 336, 399 nm.Derivative
Hydrochloride.Nomenclature
Properties
Small crystals (usually hemihydrate). Dec and turn green on exposure to light and air. [α]D25 48° (c = 1.2). uv spectrum: Csokan, Z. Anal. Chem. 124, 344 (1942). pH of aq soln (1 in 300) = 4.8. One gram dissolves in 50 ml water, 17 ml water at 80°, 50 ml alcohol. Very slightly sol in chloroform and ether. LD50 i.p. in mice: 145 μg/g (Ginos).Derivative
Diacetate (ester).Nomenclature
Properties
mp 127-128°, [α]D24 88° (c = 1.12 in 0.1N HCl).Therapeutic Category
Antiparkinsonian; emetic. In treatment of male erectile dysfunction.
Therapeutic Category (Veterinary)
Emetic.Keywords
Antiparkinsonian; Dopamine Receptor Agonist; Emetic; Impotence Therapy