748. Aporeine
Nomenclature
CAS number: 2030-53-7
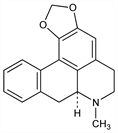
(7aS)-6,7,7a,8-Tetrahydro-7-methyl-5H-benzo[g]-1,3-benzodioxolo[6,5,4-de]quinoline; aporheine; 1,2-methylenedioxyaporphine; (+)-roemerine. C18H17NO2; mol wt 279.33.
C 77.40%, H 6.13%, N 5.01%, O 11.46%.
Description and references
From Papaver dubium L., Papaveraceae: Pavesi, Gazz. Chim. Ital. 37 I, 629 (1907); 44 I, 398 (1914). Structure and identity with (+)-roemerine: Slavik, Collect. Czech. Chem. Commun. 28, 1738 (1963). Synthesis of dl-roemerine: Marion, Grassie, J. Am. Chem. Soc. 66, 1290 (1944).
Properties
Needles from ether + petroleum ether, mp 102°. [α]D22 +80° (c = 0.50 in ethanol). uv max: 262, 315 nm (log ε 4.3, 3.7). pK 6.1. Sol in ether, methanol, ethanol, chloroform; slightly sol in petr ether. Practically insol in water, alkali.Derivative
Hydrochloride.C18H17NO2.HCl; mol wt 315.79.
C 68.46%, H 5.75%, N 4.44%, O 10.13%, Cl 11.23%.
Properties
Leaflets from ethanol or water, mp 266-267°. Slightly sol in ethanol, water.Derivative
Methiodide.C18H17NO2.CH3I; mol wt 421.27.
C 54.17%, H 4.79%, N 3.32%, O 7.60%, I 30.12%.