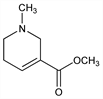
778. Arecoline
Nomenclature
Description and references
Cholinergic alkaloid from seeds of the betel nut palm Areca catechu L., Palmaceae (catechu). Isoln: E. Johns, Arch. Pharm. 229, 673 (1891). Synthesis: F. Chemnitius, J. Prakt. Chem. 117, 147 (1926); Mannich, Ber. 75B, 1480 (1942); Knox, US 2506458 (1950 to Nopco). Improved synthesis from nicotinic acid: I. A. Kozello et al., Khim. Farm. Zh. 10, 90 (1976), C.A. 86, 171205a (1977). Effect on human serial learning: N. Sitaram et al., Science 201, 274 (1978). Clinical evaluation in Alzheimer's disease: T. T. Soncrant et al., Psychopharmacology 112, 421 (1993). Review: R. B. Burrows, Progress in Drug Research vol. 17, E. Jucker, Ed. (Birkhaüser Verlag, Basel, 1973) pp 108-210.
Properties
Oily liquid, bp 209°, bp7 92-93°, bp12 105°. Volatile with steam. Strong base. pK 6.84. nD20 1.4302. d20 1.0495. Miscible with water, alc, ether. Soluble in chloroform. LD50 in mice, dogs (mg/kg): 100, 5 s.c. (Burrows).Derivative
Hydrobromide.Nomenclature
Properties
Bitter, optically inactive crystals, mp 169-171°. One gram dissolves in about 1 ml water, 10 ml alc, 2 ml boiling alc; slightly soluble in chloroform, ether. The aq soln is practically neutral.Derivative
Compd with acetarsone.Nomenclature
Properties
White to pale-yellow powder. Stable when kept dry. Freely sol in water.Derivative
Compd with p-stibonobenzoic acid.Nomenclature
Description and references
Prepn: Kartsonis, Austin, US 2557353 (1951 to Jensen-Salsbery Labs.).