784. Aricine
Nomenclature
CAS number: 482-91-7
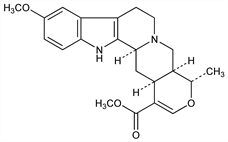
(19α,20α)-16,17-Didehydro-10-methoxy-19-methyloxayohimban-16-carboxylic
acid methyl ester; quinovatine; cinchovatine; heterophylline. C22H26N2O4; mol wt 382.45.
C 69.09%, H 6.85%, N 7.32%, O 16.73%.
Description and references
Originally found in Cusco bark, the bark of Cinchona pelletierana Wedd., Rubiaceae: Pelletier, Coriol, J. Pharm. Chim. [2] 15, 565 (1829). Also from Rauwolfia canescens L., R. heterophylla Roem. & Schult., R. sellowii Muell. Argov. and Aspidosperma marcgravianum Woodson., Apocynaceae: Stoll et al., Helv. Chim. Acta 38, 270 (1955); Hochstein et al., J. Am. Chem. Soc. 77, 3551 (1955); Hochstein, ibid. 77, 5744 (1955); Gilbert et al., J. Org. Chem. 27, 4702 (1962). Structure: Stoll et al., loc. cit. Stereochemistry: Neuss, Boaz, J. Org. Chem. 22, 1001 (1957); Shamma, Richey, J. Am. Chem. Soc. 85, 2507 (1963).
Properties
Orthorhombic, elongated prisms from methanol, dec 188°. Sublimes at 0.01 mm and 180°. [α]D20 91° (c = 1.4 in chloroform); [α]D20 63° (c = 1.5 in pyridine); [α]D20 57° (ethanol). uv max (ethanol): 229, 281 nm (log ε 4.54, 3.97). pKa in 80% methyl cellosolve 5.80; in 1:1 DMF-water 6.8. Practically insol in water. Sol in 100 parts of 90% alcohol, 33 parts ether. Very sol in chloroform.Derivative
Hydrochloride.C22H26N2O4.HCl; mol wt 418.91.
C 63.08%, H 6.50%, N 6.69%, O 15.28%, Cl 8.46%.
Properties
Square plates from methanol + acetone, dec 241-254°. [α]D20 5° (c = 0.9 in 50% ethanol).Derivative
Hydrobromide.C22H26N2O4.HBr; mol wt 463.36.
C 57.03%, H 5.87%, N 6.05%, O 13.81%, Br 17.24%.