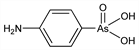
792. Arsanilic Acid
Nomenclature
Description and references
Organoarsenical originally used in treatment of trypanosomiasis. Prepd by heating aniline and arsenic acid: Lewis, Cheetham, Org. Synth. coll. vol. I (2nd ed., 1941) p 70; Hoffmann, Green, US 3763201 (1973); from p-nitroaniline: Bart, Ann. 429, 96 (1922); Gattermann-Wieland, Praxis des Organischen Chemikers (de Gruyter, Berlin, 40th ed., 1961) p 254. Metabolism in chickens: L. R. Overby, L. Straube, Toxicol. Appl. Pharmacol. 7, 850 (1965). Toxicology in swine: Ledet et al., Clin. Toxicol. 6, 439 (1973). Review of toxicology and human exposure: Toxicological Profile for Arsenic (PB2000-108021, 2000) 468 pp. Spectrophotometric determn in surface water: A. R. Roerdink, J. H. Aldstadt III, Anal. Chim. Acta 539, 181 (2005). Use as a feed additive in Japanese quail: Q. Desheng, Z. Niya, Poult. Sci. 85, 2097 (2006).