835. Asoxime Chloride
Nomenclature
Description and references
Cholinesterase reactivator. Prepn: I. Hagedorn, US 3773775 (1973 to E. Merck); L. Y. Y. Hsiao, H. A. Musallam, US 5130438 (1992 to U.S.A. Secy. of Army). Chemical stability study: P. Eyer et al., Arch. Toxicol. 59, 266 (1986). Dissolution properties: H. Thiermann et al., Int. J. Pharm. 137, 167 (1996). HPLC determn in plasma: M. P. McCluskey et al., J. Anal. Toxicol. 14, 239 (1990). Metabolism study: D. J. Ecobichon et al., Can. J. Physiol. Pharmacol. 68, 614 (1990). Clinical pharmacokinetics and safety: J. G. Clement et al., Biopharm. Drug Dispos. 16, 415 (1995). Clinical evaluation in insecticide poisoning: R. Kusic et al., Hum. Exp. Toxicol. 10, 113 (1991). Review of pharmacology: C. G. Rousseaux, A. K. Dua, Can. J. Physiol. Pharmacol. 67, 1183-1189 (1989); of physical properties and acute toxicity: J. G. Clement et al., Arch. Toxicol. 62, 220-223 (1988).
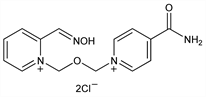
Derivative
Monohydrate.Nomenclature
Properties
Crystals from water/ethanol (20:75), mp 145-147°. uv max (H2O): 350, 300, 270, 218 nm (log ε 3.093, 4.018, 3.966, 4.181).Derivative
Dimethanesulfonate.Nomenclature
Description and references
Comparative studies of in vitro reactivation: S. Krummer et al., Arch. Toxicol. 76, 589 (2002); of in vivo pharmacokinetics and efficacy: P. M. Lundy et al., Toxicology 208, 399 (2005).
Therapeutic Category
Antidote (organophosphate poisoning).
Keywords
Antidote (Organophosphate Poisoning); Cholinesterase Reactivator