Nomenclature
CAS number: 93413-04-8
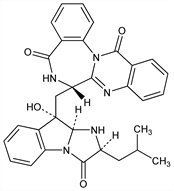
(7
S)-6,7-Dihydro-7-[[(2
S,9
S,9a
S)-2,3,9,9a-tetrahydro-9-hydroxy-2-(2-methylpropyl)-3-oxo-1
H-imidazo[1,2-
a]indol-9-yl]methyl]quinazolino[3,2-
a][1,4]benzodiazepine-5,13-dione.
C
31H
29N
5O
4; mol wt 535.59.
C 69.52%, H 5.46%, N 13.08%, O 11.95%.
Description and references
Naturally occurring, nonpeptide cholecystokinin
(CCK) antagonist. Produced by several strains of Aspergillus
alliaceus along with minor related compounds known as asperlicin
B, C, D and E. Isoln from fermentation cultures of A. alliaceus Thom and Church, ATCC 20655 and ATCC 20656: R. L. Monaghan et al., EP 116150; eidem, US 4530790 (1984, 1985 both to Merck &
Co.). Fermentation, isoln and bioactivity: M. A.
Goetz et al., J. Antibiot. 38, 1633 (1985); of asperlicins B, C, D and E: eidem, ibid. 41, 875 (1988). Structure elucidation: J. M. Liesch et al., ibid. 38, 1638 (1985);
of asperlicins B, C, D and E: eidem, ibid. 41, 878 (1988). Biosynthetic study:
D. R. Houck et al., ibid. 882. Total synthesis
of asperlicins C and E: M. G. Bock et al., J. Org. Chem. 52, 1644 (1987).
Pharmacology of asperlicin: R. S. L. Chang et al., Science 230, 177 (1985). Effect
on pancreatic enzyme secretion in vitro: K. A. Zucker et al., Surgery 102, 163 (1987). Effect in exptl pancreatitis in rats: J. R. Wisner,
Jr., I. G. Renner, Pancreas 3, 174 (1988).
Properties
White crystals, mp 211-213°. [α]D26.5 185.3° (c = 1.10 in
methanol). uv max (methanol): 310.5 nm (ε 4075). Sol
in methylene chloride, acetone and lower alcohols. Insol in water.Derivative
Asperlicin B.
Nomenclature
CAS number: 93413-08-2
C
31H
29N
5O
5; mol wt 551.59.
C 67.50%, H 5.30%, N 12.70%, O 14.50%.
Properties
Colorless powder. uv max
(methanol): 310 nm (ε 4000).Derivative
Asperlicin C.
Nomenclature
CAS number: 93413-06-0
C
25H
18N
4O
2; mol wt 406.44.
C 73.88%, H 4.46%, N 13.78%, O 7.87%.
Properties
Off-white powder. uv max
(methanol): 222, 268, 278, 310 nm (ε 56300,
15100, 14000, 4650).Derivative
Asperlicin D.
Nomenclature
CAS number: 93413-07-1
C
25H
18N
4O
2; mol wt 406.44.
C 73.88%, H 4.46%, N 13.78%, O 7.87%.
Properties
Colorless powder. uv max
(methanol): 222, 290, 310 nm (ε 61060, 14600, 5920).Derivative
Asperlicin E.
Nomenclature
CAS number: 93413-05-9
C
25H
18N
4O
3; mol wt 422.44.
C 71.08%, H 4.29%, N 13.26%, O 11.36%.
Properties
Off-white powder. uv max
(methanol): 227, 268, 324 nm (ε 38450, 8680, 3200).