851. Aspirin
Nomenclature
Description and references
Prepn: C. Gerhardt, Ann. 87, 149 (1853). Manuf from salicylic acid and acetic anhydride: Faith, Keyes & Clark's Industrial Chemicals, F. A. Lowenheim, M. K. Moran, Eds. (Wiley-Interscience, New York, 4th ed., 1975) pp 117-120. Crystallization from acetone: Hamer, Phillips, US 2890240 (1959 to Monsanto). Novel process involving distillation: Edmunds, US 3235583 (1966 to Norwich Pharm.). Crystal structure: P. J. Wheatley, J. Chem. Soc. (Suppl.) 1964, 6036. Characterization of a second polymorphic form: P. Vishweshwar et al., J. Am. Chem. Soc. 127, 16802 (2005). Toxicity data: E. R. Hart, J. Pharmacol. Exp. Ther. 89, 205 (1947). Evaluation as a risk factor in Reye's syndrome: P. J. Waldman et al., J. Am. Med. Assoc. 247, 3089 (1982). Review of clinical trials in prevention of myocardial infarction and stroke: P. C. Elwood, Drugs 28, 1-5 (1984). Symposium on aspirin therapy: Am. J. Med. 74, no. 6A, 1-109 (1983). Clinical trials in prevention of colorectal cancer: R. S. Sandler et al., N. Engl. J. Med. 348, 883 (2003); J. A. Baron et al., ibid. 891. Comprehensive description: K. Florey, Anal. Profiles Drug Subs. 8, 1-46 (1979). Monograph: M. J. H. Smith, P. K. Smith, The Salicylates (Interscience, New York, 1966) 313 pp. Book: Acetylsalicylic Acid, H. J. M. Barnett et al., Eds. (Raven, New York, 1982) 278 pp.
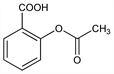
Properties
White monoclinic tablets or needle-like crystals. d 1.40. mp 135° (rapid heating); the melt solidifies at 118°. uv max (0.1N H2SO4): 229 nm (E1%1cm 484); (CHCl3): 277 nm (E1%1cm 68). Is odorless, but in moist air it is gradually hydrolyzed into salicylic and acetic acids and acquires the odor of acetic acid. Stable in dry air. pK (25°) 3.49. One gram dissolves in 300 ml water at 25°, in 100 ml water at 37°, in 5 ml alcohol, 17 ml chloroform, 10-15 ml ether. Less soluble in anhydr ether. Decomp by boiling water or when dissolved in solns of alkali hydroxides and carbonates. Inorganic salts of acetylsalicylic acid are soluble in water (esp the Ca salt, q.v.), but are decomposed quickly. LD50 orally in mice, rats: 1.1, 1.5 g/kg (Hart).Derivative
Guaiacol ester.Nomenclature
Derivative
Methyl ester see Methyl Acetylsalicylate.Derivative
Phenyl ester see Phenyl Acetylsalicylate.Caution
Potential symptoms of overexposure are increased blood clotting time; nausea, vomiting; liver, kidney injury. Direct contact may cause eye, skin and upper respiratory irritation. See NIOSH Pocket Guide to Chemical Hazards (DHHS/NIOSH 97-140, 1997) p 6.Therapeutic Category
Analgesic; antipyretic; anti-inflammatory; antithrombotic.
Therapeutic Category (Veterinary)
Analgesic; antipyretic; anti-inflammatory; antithrombotic.Keywords
Anti-inflammatory (Nonsteroidal); Salicylic Acid Derivatives; Antipyretic; Analgesic; Non-opioids; Salicylic Acid Derivatives; Antithrombotic