Nomenclature
CAS number: 514-76-1
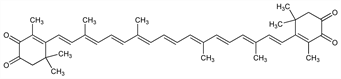
β,β-Carotene-3,3′,4,4′-tetrone; 3,4,3′,4′-tetraketo-β-carotene; 3,3′-dihydroxy-2,3,2′,3′-tetradehydro-β,β-carotene-4,4′-dione; astacene.
C
40H
48O
4; mol wt 592.81.
C 81.04%, H 8.16%, O 10.80%.
Description and references
Red carotenoid pigment isolated from biological
material originating from crustacea, algae, sponges, protozoa, fish
and reptiles. Small amounts were isolated from the fat of mammals
(whales, Balaenoptera musculus). Occurs together with
astaxanthin from which it is formed by autoxidation. Appears to be
an artifact rather than a natural product. Isoln from lobster shells:
Kuhn, Lederer, Ber. 66, 488 (1933). Structure: Karrer et al., Helv. Chim. Acta 17, 412, 745
(1934); 18, 96 (1935); 19, 479 (1936). Total
synthesis: J. B. Davis, B. C. L. Weedon, Proc. Chem. Soc. London 1960, 182; E. Widmer et al., Helv. Chim. Acta 65, 671 (1982). Prepn by autoxidation of canthaxanthin:
R. D. G. Cooper et al., J. Chem.
Soc. Perkin Trans. 1 1975, 2195.
Properties
Purple needles or leaflets with metallic luster,
sometimes bent into sickle shape, esp when crystallized from pyridine
+ water, mp 240-243° (slow
heating in evac tube, Karrer), mp 228° (Kuhn). Absorption
max (pyridine): 500 nm. Practically insol in water; freely sol in chloroform, pyridine,
dioxane, carbon disulfide and dil aq alkali; slightly sol in benzene,
ethyl acetate, glacial acetic acid; nearly insol in ether, petr ether,
methanol.Derivative
Diacetate.
C
44H
52O
6; mol wt 676.88.
C 78.07%, H 7.74%, O 14.18%.
Properties
Black to violet needles from pyridine + water,
dec 235°.Derivative
Dipalmitate (astacein).
C
72H
108O
6; mol wt 1069.62.
C 80.85%, H 10.18%, O 8.97%.
Properties
Almost square red leaflets from petr ether, mp 121°.