875. Atropine
Nomenclature
Description and references
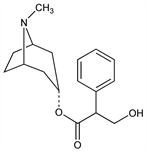
Parasympatholytic alkaloid isolated from Atropa belladonna L., Datura stramonium L., and other Solanaceae. Extraction procedure: Chemnitius, J. Prakt. Chem. 116, 276 (1927). During extraction, partial racemization of the l-hyoscyamine takes place which is completed by treatment with dil alkali on heating in chloroform soln: Schneider, Arch. Pharm. 284, 306 (1951). Structure and synthesis: Ladenburg, Ann. 217, 75 (1883); Willsttter, Ber. 31, 1537 (1898); idem., Ann. 326, 23 (1903); Schwenker et al., Ber. 99, 2407 (1966). Prepn of the sulfate: DE 247455 (1912 to Hoffmann-La Roche), Frdl. 11, 1022. Use as antidote to cholinesterase inhibitors: R. V. Brown, Br. J. Pharmacol. 15, 170 (1960). Effect on cardiac arrhythmias: P. Schweitzer, H. Mark, Am. Heart J. 100, 119, 255 (1980). Pharmacokinetics and pharmacodynamics: P. H. Hinderling et al., J. Pharm. Sci. 74, 703, 711 (1985). Toxicity: R. L. Cahen, K. Tvede, J. Pharmacol. Exp. Ther. 105, 166 (1952); Goldenthal, Toxicol. Appl. Pharmacol. 18, 185 (1971). Review of clinical use in anesthesia: L. E. Shutt, J. B. Bowes, Anaesthesia 34, 476-490 (1979). Comprehensive description: A. A. Al-Badr, F. J. Muhtadi, Anal. Profiles Drug Subs. 14, 325-389 (1985). Review of clinical toxicology: J. D. Truwit, Crit. Care Clin. 7, 639-657 (1991).
Properties
Long, orthorhombic prisms from acetone, mp 114-116°. Sublimes in high vacuum at 93-110°. pK 4.35; pH of 0.0015 molar soln 10.0. Absorption spectra: Dobbie, Fox, J. Chem. Soc. 103, 1194 (1913); Fischer, Arch. Exp. Pathol. Pharmakol. 170, 623 (1933). One gram dissolves in 455 ml water, 90 ml water at 80°, 2 ml alc, 1.2 ml alc at 60°, 27 ml glycerol, 25 ml ether, 1 ml chloroform; also sol in benzene, dil acids. LD50 orally in rats: 750 mg/kg (Cahen, Tvede).Derivative
Methylbromide.Nomenclature
Properties
Crystals, mp 222-223°. Sol in 1 part water; slightly sol in alc. Almost insol in chloroform, ether.Derivative
Methylnitrate.Nomenclature
Properties
Crystals, mp 163°. Freely sol in water or alc, very slightly in chloroform, ether.Derivative
Sulfate monohydrate.Nomenclature
Properties
Granules or powder, mp 190-194°. Almost inactive optically. Very bitter. pH ≈5.4. One gram dissolves in 0.4 ml water; 5 ml cold, 2.5 ml boil. alc; in 2.5 ml glycerol, 420 ml chloroform, 3000 ml ether. Bitterness threshold 1:10,000. Incompatible with alkalies, tannin, salts of mercury or gold, vegetable decoctions or infusions, borax, bromides, iodides, benzoates. LD50 orally in rats: 622 mg/kg (Goldenthal).Derivative
N-Oxide.Nomenclature
Description and references
Prepn: Polonovski, Polonovski, Bull. Soc. Chim. Fr. 39, 1147 (1926).
Properties
Crystalline powder, mp 127-128°, dec 135°. Very hygroscopic. Soluble in alc, chloroform. Practically insol in ether.Caution
Causes blurred vision, suppressed salivation, vasodilation, hyperpyrexia, excitement, agitation, and delirium. See: Clinical Toxicology of Commercial Products, R. E. Gosselin et al., Eds. (Williams & Wilkins, Baltimore, 5th Ed., 1984) Section III, pp 47-50.Therapeutic Category
Mydriatic; antispasmodic. In preanesthetic medication.
Therapeutic Category (Veterinary)
Mydriatic; antispasmodic. Antidote to organophosphorus insecticides.Keywords
Mydriatic; Antimuscarinic; Antispasmodic