904. 6-Azauridine
Nomenclature
Description and references
Cytostatic nucleoside analog; inhibits orotidyl decarboxylase and blocks de novo pyrimidine biosynthesis. Production by E. coli in the presence of 6-azauracil: J. Skoda et al., Experientia 13, 150 (1957). Chemical synthesis: M. Prystas et al., Chem. Ind. (London) 1961, 947. Crystal structure and conformation: C. H. Schwalbe et al., Biochem. Biophys. Res. Commun. 44, 57 (1971). Degradation kinetics: C. M. Riley et al., Pharm. Res. 12, 1361 (1995). Clinical evaluation in recalcitrant psoriasis: J. C. Alper et al., J. Am. Acad. Dermatol. 13, 567 (1985). Toxicity study: J. Plevova et al., Toxicol. Appl. Pharmacol. 17, 511 (1970). Review of effect on vitamin B6 and homocystinemia: W. Drell, A. D. Welch, Pharmacol. Ther. 41, 195-206 (1989).
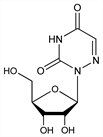
Properties
Crystals from ethanol, ether, mp 160-161°. [α]D24 132° (pyridine). uv max (water): 262 nm (ε 6100). pK 6.70.Derivative
2′,3′,5′-Triacetate.Nomenclature
Properties
pKa (25°): 6.42. LD50 in mice, rats (g/kg): 7.8, 12.0 orally (Plevova).Therapeutic Category
Antipsoriatic.
Keywords
Antineoplastic; Antimetabolites; Pyrimidine Analogs; Antipsoriatic