958. Baptigenin
Nomenclature
CAS number: 5908-63-4
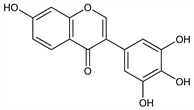
7-Hydroxy-3-(3,4,5-trihydroxyphenyl)-4H-1-benzopyran-4-one; 7,3′,4′,5′-tetrahydroxyisoflavone. C15H10O6; mol wt 286.24.
C 62.94%, H 3.52%, O 33.54%.
Description and references
The aglucon of baptisin. From baptisin by acid hydrolysis or vacuum sublimation. From radix Baptisiae: Fischer, Ehrlich, C.A. 31, 44493 (1937). Structure: Bhm, Arzneim.-Forsch. 10, 472 (1960). Synthesis: Farkas et al., Ber. 96, 1865 (1963). See also Pseudobaptigenin.
Properties
Needles from dil ethanol, mp 284-285°. Begins to sublime at 240°. Sublimes in oil pump vacuum at 180-200°. uv max (ethanol): 270.2, 247 nm. Practically insol in water, ammonia water; slightly sol in dil alc, hot glacial acetic acid; sol in acetone, in NaOH solns.Derivative
Tetraacetylbaptigenin.C23H18O10; mol wt 454.38.
C 60.80%, H 3.99%, O 35.21%.
Properties
Needles from methanol, mp 214°.Derivative
Tetrabenzoylbaptigenin.C43H26O10; mol wt 702.66.
C 73.50%, H 3.73%, O 22.77%.
Properties
Prisms from methanol, mp 191-192°.Derivative
Tetramethylbaptigenin.C19H18O6; mol wt 342.34.
C 66.66%, H 5.30%, O 28.04%.