1029. Benactyzine
Nomenclature
CAS number: 302-40-9
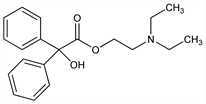
α-Hydroxy-α-phenylbenzeneacetic acid 2-(diethylamino)ethyl
ester; benzilic acid β-diethylaminoethyl ester; β-diethylaminoethyl benzilate; 2-diethylaminoethyl diphenylglycolate. C20H25NO3; mol wt 327.42.
C 73.37%, H 7.70%, N 4.28%, O 14.66%.
Description and references
An antagonist of acetylcholine in the central and peripheral nervous systems. Prepn: Horenstein, Pahlicke, Ber. 71, 1654 (1938); Blicke, Maxwell, J. Am. Chem. Soc. 64, 428 (1942); Hill, Holmes, US 2394770 (1946 to Am. Cyanamid). Toxicity and pharmacodynamics: Fournier, Petit, Therapie 17, 1245 (1962). Metabolism: Eldeson et al., Arch. Int. Pharmacodyn. Ther. 187, 139 (1970). Crystal and molecular structure determn by x-ray diffraction: T. J. Petcher, J. Chem. Soc. Perkin Trans. 2 1974, 1151.
Properties
Crystals, mp 51°.Derivative
Hydrochloride.Nomenclature
CAS number: 57-37-4
AY-5406-1; Cedad (Recordati); Nutinal (Boots); Parasan; Suavitil (Merck & Co.). C20H25NO3.HCl; mol wt 363.88.
C 66.01%, H 7.20%, N 3.85%, O 13.19%, Cl 9.74%.
Properties
Crystals from acetone, mp 177-178°. Soly in water (25°): 14.9/100 ml. Practically insol in ether.Derivative
Methobromide.Nomenclature
CAS number: 3166-62-9
Spatomac. C20H25NO3.CH3Br; mol wt 422.36.
C 59.72%, H 6.68%, N 3.32%, O 11.36%, Br 18.92%.
Properties
Crystals from alcohol + ether, mp 169-170°.Therapeutic Category
Antispasmodic.
Keywords
Antimuscarinic; Antispasmodic