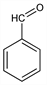
1058. Benzaldehyde
Nomenclature
CAS number: 100-52-7
Benzoic aldehyde; artificial essential oil of almond. C7H6O; mol wt 106.12.
C 79.23%, H 5.70%, O 15.08%.
Description and references
Occurs in kernels of bitter almonds; made synthetically from benzal chloride and lime or by oxidation of toluene. Laboratory prepn from benzal chloride: A. I. Vogel, Practical Organic Chemistry (Longmans, London, 3rd ed., 1959) p 693; Gattermann-Wieland, Praxis des Organischen Chemikers (de Gruyter, Berlin, 40th ed., 1961) p 184. Toxicity data: P. M. Jenner et al., Food Cosmet. Toxicol. 2, 327 (1964). Review: A. E. Williams in Kirk-Othmer Encyclopedia of Chemical Technology vol. 3 (Wiley-Interscience, New York, 3rd ed., 1978) pp 736-743.