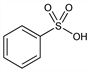
1070. Benzenesulfonic Acid
Nomenclature
CAS number: 98-11-3
C6H6O3S; mol wt 158.18.
C 45.56%, H 3.82%, O 30.34%, S 20.27%.
Description and references
Made by treating benzene with fuming H2SO4: Gattermann-Wieland, Praxis des Organischen Chemikers (de Gruyter, Berlin, 40th ed., 1961) p 168.
Derivative
Sesquihydrate.Nomenclature
CAS number: 5928-72-3
C6H6O3S.1H2O; mol wt 167.18.
C 43.11%, H 4.22%, O 33.50%, S 19.18%.
Properties
Deliquescent plates, tablets, mp 43-44°. When anhydrous mp 50-51°, also reported as mp 65-66°. pKa (25°): 0.699. Freely sol in water, alc; slightly sol in benzene; insol in ether, carbon disulfide. Keep well closed.Derivative
Ethyl ester.Nomenclature
CAS number: 515-46-8
Ethyl benzenesulfonate. C8H10O3S; mol wt 186.23.
C 51.60%, H 5.41%, O 25.77%, S 17.22%.
Properties
Colorless to slightly yellow, almost odorless liq. bp15 156°. d417 1.219. Slightly sol in water; misc with alc, benzene, chloroform, ether.Derivative
Sodium salt.Nomenclature
CAS number: 515-42-4
Sodium benzenesulfonate; sodium benzosulfonate. C6H5NaO3S; mol wt 180.16.
C 40.00%, H 2.80%, Na 12.76%, O 26.64%, S 17.80%.