1098. Benzophenone
Nomenclature
CAS number: 119-61-9
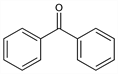
Diphenylmethanone; diphenyl ketone; benzoylbenzene. C13H10O; mol wt 182.22.
C 85.69%, H 5.53%, O 8.78%.
Description and references
Prepd by the Friedel-Crafts ketone synthesis from benzene and benzoyl chloride in the presence of AlCl3: Marvel, Sperry, Org. Synth. coll. vol. I (Wiley, New York, 2nd ed., 1941) p 95. By decarboxylation of o-benzoylbenzoic acid in the presence of copper catalyst: L. F. Fieser, Organic Experiments (D. C. Heath & Co., Boston, 1964) pp 201-203.
Properties
Stable form (there are two other labile forms), orthorhombic bisphenoidal prisms from alcohol or ether. Geranium-like odor. d418 1.1108. d450 1.0869. nD45.2 1.5975. Absorption spectrum: Purvis, McCleland, J. Chem. Soc. 101, 1516 (1912). mp 48.5°. bp760 305.4°; bp400 276.8°; bp200 249.8°; bp100 224.4°; bp60 208.2°; bp40 195.7°; bp20 175.8°; bp10 157.6°; bp5 141.7°; bp1.0 108.2°. Insoluble in water. One gram dissolves in 7.5 ml alcohol, 6 ml ether; sol in chloroform.Derivative
Oxime.Nomenclature
CAS number: 574-66-3
C13H11NO; mol wt 197.23.
C 79.17%, H 5.62%, N 7.10%, O 8.11%.