1180. Betamethasone
Nomenclature
CAS number: 378-44-9
(11β,16β)-9-Fluoro-11,17,21-trihydroxy-16-methylpregna-1,4-diene-3,20-dione; 9α-fluoro-16β-methylprednisolone; 16β-methyl-9α-fluoro-Δ1-hydrocortisone; 16β-methyl-9α-fluoroprednisolone; betadexamethasone; flubenisolone; β-methasone; Sch-4831; NSC-39470; beta-Corlan; Becort (Rachelle); Betasolon; Betnelan (GSK); Celestene (Schering-Plough); Celestone (Schering-Plough); Dermabet (Taro); Visubeta. C22H29FO5; mol wt 392.46.
C 67.33%, H 7.45%, F 4.84%, O 20.38%.
Description and references
Prepn: Taub et al., J. Am. Chem. Soc. 80, 4435 (1958); Oliveto et al., ibid. 6688; Taub et al., ibid. 82, 4012 (1960); US 3053865 (1962 to Merck & Co.); Amiard et al., US 3104246 (1963 to Roussel-UCLAF). Also prepared from hecogenin. Comprehensive description of the dipropionate ester: M. G. Ferrante, B. C. Rudy, Anal. Profiles Drug Subs. 6, 43-60 (1977).
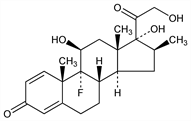
Properties
Crystals from ethyl acetate, mp 231-234° (dec). [α]D +108° (acetone). uv max (methanol): 238 nm (ε 15200).Derivative
21-Acetate.Nomenclature
CAS number: 987-24-6
Betafluorene (Lepetit); Celestovet (Biokema). C24H31FO6; mol wt 434.50.
C 66.34%, H 7.19%, F 4.37%, O 22.09%.
Properties
Hexagonal prisms from acetone + ether, mp 205-208° (Taub); also reported as mp 196-201° (Oliveto). [α]D +140° (chloroform). uv max (methanol): 238 nm (ε 14800).Derivative
17-Benzoate.Nomenclature
CAS number: 22298-29-9
W-5975; Bebate; Beben (Pfizer); Benisone (Warner-Chilcott); Euvaderm (G"odecke); Flurobate (Texas Pharmacal); Uticort (Pfizer). C29H33FO6; mol wt 496.57.
C 70.14%, H 6.70%, F 3.83%, O 19.33%.
Properties
Crystals from acetone-ether, mp 225-228°. [α]D24 +63.5° (dioxane). Synthesis and activity: Ercoli et al., J. Med. Chem. 15, 783 (1972). See also Cullen, Curr. Ther. Res. 15, 243 (1973).Derivative
17,21-Dipropionate.Nomenclature
CAS number: 5593-20-4
Sch-11460; Diproderm (Schering-Plough); Diprolene (Schering-Plough); Diprophos (Schering-Plough); Diprosis (Essex); Diprosone (Schering-Plough); Maxivate (Westwood); Rinderon-DP (Shionogi). C28H37FO7; mol wt 504.59.
C 66.65%, H 7.39%, F 3.77%, O 22.20%.
Properties
Powder, mp 170-179° (dec). [α]D26 +65.7° (dioxane). uv max (methanol): 238 nm (ε 15700). Insol in water.Derivative
17-Valerate.Nomenclature
CAS number: 2152-44-5
Bedermin (Damor); Betnesol-V (GSK); Betneval (GSK); Betnovate (GSK); Bextasol (GSK); Celestan-V (Essex); Celestoderm-V (Schering-Plough); Dermosol (Iwaki); Dermovaleas (Valeas); Ecoval 70; Hormezon (Tobishi); Tokuderm (Nichiban); Valisone (Schering-Plough). C27H37FO6; mol wt 476.58.
C 68.05%, H 7.83%, F 3.99%, O 20.14%.
Properties
Needles from acetone + petr ether, mp 183-184°. [α]D +77° (dioxane). uv max: 239 nm (ε 15920). NL 6406615 (1964 to Glaxo).Derivative
21-Phosphate disodium salt.Nomenclature
CAS number: 151-73-5
Betamethasone 21-(dihydrogen phosphate) disodium salt; Bentelan (GSK); Betnesol (GSK); Celestan (Essex); Durabetason (Durachemie); Vista-Methasone (Richard Daniel). C22H28FNa2O8P; mol wt 516.40.
C 51.17%, H 5.47%, F 3.68%, Na 8.90%, O 24.79%, P 6.00%.
Therapeutic Category
Glucocorticoid.
Therapeutic Category (Veterinary)
Glucocorticoid.Keywords
Glucocorticoid