1189. Betulin
Nomenclature
CAS number: 473-98-3
(3β)-Lup-20(29)-ene-3,28-diol; lup-20(30)-ene-3β,28-diol; trochol; betulinol; betulol. C30H50O2; mol wt 442.72.
C 81.39%, H 11.38%, O 7.23%.
Description and references
In the outer portion of the bark of white birch (up to 24%), in other barks, and in lignite. Isoln: Lowitz, Crell's Chem. Ann. 1, 312 (1788); L. Ruzicka, O. Isler, Helv. Chim. Acta 19, 506 (1936); and botanical distribution: Steiner, Molisch Festschrift (1936). Isoln from Lemaireocereus griseus Britton et Rose, Cactaceae: C. Djerassi et al., J. Am. Chem. Soc. 78, 2312 (1956). Structure: Ames et al., J. Chem. Soc. 1951, 450; Davy et al., ibid. 1951, 2696, 2702. Stereochemistry: Guider et al., ibid. 1953, 3024; Das, Chem. Ind. (London) 1971, 1331. Reviews: J. Simonsen, W. C. J. Ross, The Terpenes vol. IV (Cambridge Univ. Press, 1957) pp 187-328; E. W. H. Hayek et al., Phytochemistry 28, 2229-2242 (1989).
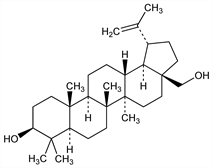
Properties
Crystals from methanol-chloroform, mp 248-251°; sublimes at 240° at 0.01 mm. Solvated needles from alc contg one mol EtOH. After drying sublimes at 170-180° (bath temp) at 0.08 mm. uv max (H2SO4): 316 nm. [α]D15 +20° (c = 2 in pyridine). Sparingly sol in cold water, petr ether, carbon disulfide. One part is sol in 149 parts alc, 251 ether, 113 chloroform, 417 benzene. Sol in acetic acid.Derivative
Diacetate.Nomenclature
CAS number: 1721-69-3
C34H54O4; mol wt 526.79.
C 77.52%, H 10.33%, O 12.15%.