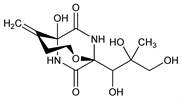
1202. Bicyclomycin
Nomenclature
Description and references
Structurally unique antibiotic; exhibits broad spectrum activity vs Gram-negative bacteria and the Gram-positive bacterium, Micrococcus luteus. Prepn by fermentation of Streptomyces sapporensis: T. Miyoshi et al., DE 2150593; H. Imanaka et al., US 3923790 (1972, 1975 both to Fujisawa). Isoln and characterization: T. Miyoshi et al., J. Antibiot. 25, 569 (1972). Isoln from S. aizunensis and identity with antibiotic 5879: S. Miyamura et al., ibid. 26, 479 (1973). Structural elucidation: T. Kamiya et al., ibid. 25, 576 (1972). Crystal and molecular structure: Y. Tokuma et al., Bull. Chem. Soc. Jpn. 47, 18 (1974). In vitro and in vivo activity studies: M. Nishida et al., J. Antibiot. 25, 582 (1972). Metabolism: eidem, ibid. 594. Total synthesis of (±)-bicyclomycin: S. Nakatsuka et al., Tetrahedron Lett. 24, 5627 (1983); of (+)-bicyclomycin: R. M. Williams et al., J. Am. Chem. Soc. 106, 5749 (1984); eidem, ibid. 107, 3253 (1985). Efficacy vs Shiga toxin-producing E. coli in calves: N. Misawa et al., Microbiol. Immunol. 44, 891 (2000). Review of synthetic, mechanistic and biological studies: R. M. Williams, C. A. Durham, Chem. Rev. 88, 511-540 (1988); of mechanism of action: H. Kohn, W. Widger, Curr. Drug Targets Infect. Disord. 5, 273-295 (2005).