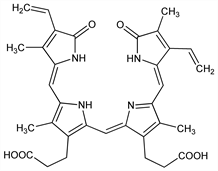
1219. Biliverdine
Nomenclature
Description and references
Precursor of bilirubin. Formed in the body from hemoglobin. The bile of amphibia and of birds contains biliverdine only. Does not occur in normal human bile or normal human serum, but regularly accompanies bilirubin in the serum of patients with carcinomatous obstruction of the bile duct, and frequently in that of patients with liver cirrhosis, catarrhal jaundice, and bile duct occlusion by gallstones. Can be obtained by autooxidation of bilirubin in alkaline soln but the yield is poor: Lemberg, Biochem. J. 28, 978 (1934); better yields by oxidation of bilirubin with ferric chloride in glacial acetic acid: Lemberg, Ann. 499, 25 (1932); by coupled oxidation of hemoglobin and ascorbic acid: Lemberg et al., Biochem. J. 35, 363 (1941); by oxidation of bilirubin with ferric chloride in methanol: Gray et al., J. Chem. Soc. 1961, 2264. Crystal and molecular structure: W. S. Sheldrick, J. Chem. Soc. Perkin Trans. 2 1976, 1457. Comprehensive reviews: Lemberg, Legge, Hematin Compounds and Bile Pigments (New York, 1949); With, Bile Pigments (Academic Press, New York, 1968).