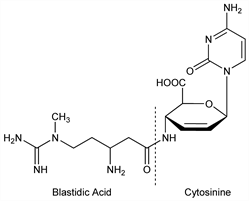
1316. Blasticidin S
Nomenclature
CAS number: 2079-00-7
4-[[3-Amino-5-[(aminoiminomethyl)methylamino]-1-oxopentyl]amino]-1-(4-amino-2-oxo-1(2H)-pyrimidinyl)-1,2,3,4-tetradeoxy-β-d-erythro-hex-2-enopyranuronic acid; 4-[3-amino-5-(1-methylguanidino)valeramido]-1-(4-amino-2-oxo-1(2H)-pyrimidinyl)-1,2,3,4-tetradeoxy-β-d-erythro-hex-2-enopyranuronic acid; 1-(1′-cytosinyl)-4-[l-3′-amino-5′-(1″-N-methylguanidino)-valerylamino]-1,2,3,4-tetradeoxy-β-d-erythro-hex-2-enuronic acid. C17H26N8O5; mol wt 422.44.
C 48.33%, H 6.20%, N 26.53%, O 18.94%.
Description and references
Nucleoside antibiotic produced by Streptomyces griseochromogenes: Isoln and antimicrobial activity: Takeuchi et al., J. Antibiot. 11A, 1 (1958); Sumiki, Umezawa, JP 60 16449 (1960 to Japan Antibiot. Res. Assoc.), C.A. 55, 21474e (1961). Structure: Otake et al., Agric. Biol. Chem. 30, 132 (1966); Fox, Watanabe, Tetrahedron Lett. 1966, 897. Abs config: Yonehara et al., ibid. 1966, 3785. Synthesis of the unsaturated carbohydrate: Goody et al., ibid. 1970, 293; of the cytosinine moiety: Fox, Watanabe, Pure Appl. Chem. 28, 475 (1971); Kondo et al., Tetrahedron Lett. 1972, 1881; eidem, Tetrahedron 29, 1801 (1973). Crystal and molecular structure: V. Swaminathan et al., Biochim. Biophys. Acta 655, 335 (1981).