1428. Bromophenol
C6H5BrO; mol wt 173.01.
C 41.65%, H 2.91%, Br 46.18%, O 9.25%.
Description and references
Prepn of m-isomer: Wurster, Nlting, Ber. 7, 904 (1874); Carpenter et al., J. Org. Chem. 16, 586 (1951); of o- and p-isomers: Medola, Streatfield, J. Chem. Soc. 73, 681 (1898); Kaeding, Lindstrom, US 2805263 (1957 to Dow).
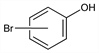
Derivative
m-Bromophenol.Nomenclature
CAS number: 591-20-8
Properties
Crystals, mp 33°, also reported as mp 31°. bp 235-236°, bp3 88-89°. Sol in alcohol, ether, alkalies.Derivative
o-Bromophenol.Nomenclature
CAS number: 95-56-7
Properties
Yellow to red oily liquid; unpleasant odor. d ≈1.5. bp 194°. mp 6°. Sol in water; miscible in chloroform, ether. Fusion with NaOH gives resorcinol.Derivative
p-Bromophenol.Nomenclature
CAS number: 106-41-2
Properties
Tetragonal bipyramidal crystals from chloroform or ether. mp 64°. bp 238°. d15 1.840; d80 1.5875. Small amounts of water depress the mp considerably and may prevent crystallization. Absorption spectrum: Ley, Z. Phys. Chem. 94, 412 (1920). Soluble in about 7 parts water; freely sol in alc, chloroform, ether, glacial acetic acid.Derivative
p-Form methyl ether.Nomenclature
p-Bromoanisole.C7H7BrO; mol wt 187.03.
C 44.95%, H 3.77%, Br 42.72%, O 8.55%.
Properties
Crystals, mp 9-10°. bp 223°.Derivative
p-Form ethyl ether.Nomenclature
p-Bromophenetole.C8H9BrO; mol wt 201.06.
C 47.79%, H 4.51%, Br 39.74%, O 7.96%.