1593. Butyric Acid
Nomenclature
CAS number: 107-92-6
Butanoic acid; n-butyric acid; ethylacetic acid. C4H8O2; mol wt 88.11.
C 54.53%, H 9.15%, O 36.32%.
Description and references
Discovered by Lieben and Rossi in 1869. Present in butter as an ester to the extent of 4-5%. Obtained by suitable fermentation of carbohydrates; prepn from n-propanol + CO at 200 atm in the presence of Ni(CO)4 and NiI2: Reppe et al., Ann. 582, 83 (1953); lab prepn from ethylmalonic acid: Gattermann-Wieland, Praxis des Organischen Chemikers (de Gruyter, Berlin, 40th ed., 1961) p 221. Toxicity study: Smyth et al., Arch. Ind. Hyg. Occup. Med. 10, 61 (1954).
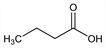
Properties
Oily liq; unpleasant, rancid odor. d420 0.959. bp 163.5°. mp -7.9°. nD20 1.3991. Flash pt, closed cup: 170°F (77°C). Corrosive. Neutralization value 636.79. Miscible with water, alcohol, ether. The calcium salt of this acid is less soluble in hot than in cold water (difference from isobutyric acid). LD50 orally in rats: 8.79 g/kg (Smyth).Derivative
Magnesium salt.Nomenclature
CAS number: 556-45-6
Magnesium butyrate. C8H14MgO4; mol wt 198.50.
C 48.41%, H 7.11%, Mg 12.24%, O 32.24%.