1600. Cabenegrins
Description and references
Orally active antidotes against snake venoms; isolated from the root of a South American plant called “Cabeca de Negra” and structurally related to pterocarpin, q.v. Isoln: L. L. Darko et al., EP 89229; eidem, US 4429141 (1983, 1984 both to Richter, Budapest). Structure determn: M. Nakagawa et al., Tetrahedron Lett. 1982, 3855. Synthesis of (±)-cabenegrins A-I and A-II: M. Ishiguro et al., ibid. 3859.
Derivative
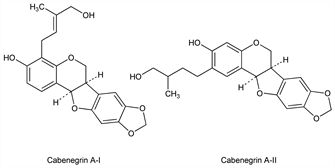
Cabenegrin A-I.Nomenclature
CAS number: 84297-59-6
[6aR-[4(E),6aα,12aα]]-6a,12a-Dihydro-4-(4-hydroxy-3-methyl-2-butenyl)-6H-[1,3]dioxolo[5,6]benzofuro[3,2-c][1]benzopyran-3-ol. C21H20O6; mol wt 368.38.
C 68.47%, H 5.47%, O 26.06%.
Properties
White crystalline solid, mp 167-168°. uv max (ethanol): 309 nm (ε 13000); uv max (methanol): 209, 233, 309 nm (ε 75000, 24000, 13000).Derivative
Cabenegrin A-II.Nomenclature
CAS number: 84297-60-9
6a,12a-Dihydro-3-hydroxy-β-methyl-6H-[1,3]dioxolo[5,6]benzofuro[3,2-c][1]benzopyran-2-butanol. C21H22O6; mol wt 370.40.
C 68.10%, H 5.99%, O 25.92%.