1636. Caffeine
Nomenclature
Description and references
Naturally occurring alkaloid commonly found in tea leaves, coffee beans, cocoa beans, maté leaves, guarana paste and kola nuts. Originally isolated from coffee in 1821. Synthesis: E. Fischer, Ber. 15, 453 (1882); E. Fischer, L. Ach, ibid. 28, 3135 (1895); W. Traube, ibid. 33, 1371, 3035 (1900); H. Bredereck et al., ibid. 83, 201 (1950). Crystal structure: D. J. Sutor, Acta Crystallogr. 11, 453 (1958). Arrhythmogenic effects in humans: D. J. Dobmeyer et al., N. Engl. J. Med. 308, 814 (1983). Teratogenicity study: P. E. Palm et al., Toxicol. Appl. Pharmacol. 44, 1 (1978). HPLC determn in serum: D. T. Holland et al., J. Chromatogr. B 707, 105 (1998). Comprehensive description: M. U. Zubair et al., Anal. Profiles Drug Subs. 15, 71-150 (1986). Review of dietary sources: D. M. Graham, Nutr. Rev. 36, 97-102 (1978); of clinical pharmacology: N. L. Benowitz, Annu. Rev. Med. 41, 277-288 (1990); of CNS effects: A. Nehlig et al., Brain Res. Rev. 17, 139-170 (1992); of therapeutic uses: J. Sawynok, Drugs 49, 37-50 (1996).
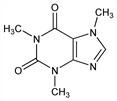
Properties
Hexagonal prisms by sublimation, mp 238°. Odorless with bitter taste. Sublimes 178°. Fast sublimation is obtained at 160-165° under 1 mm press. at 5 mm distance. d418 1.23. pH of 1% soln 6.9. Absorption spectrum: Hartley, J. Chem. Soc. 87, 1802 (1905). One gram dissolves in 46 ml water, 5.5 ml water at 80°, 1.5 ml boiling water, 66 ml alcohol, 22 ml alcohol at 60°, 50 ml acetone, 5.5 ml chloroform, 530 ml ether, 100 ml benzene, 22 ml boiling benzene. Freely sol in pyrrole; in tetrahydrofuran contg about 4% water; also sol in ethyl acetate; slightly in petr ether. Soly in water is increased by alkali benzoates, cinnamates, citrates or salicylates. LD50 orally in mice, hamsters, rats, rabbits (mg/kg): 127, 230, 355, 246 (males); 137, 249, 247, 224 (females) (Palm).Derivative
Monohydrate.Properties
Felted needles, contg 8.5% H2O. Efflorescent in air; complete dehydration takes place at 80°.Derivative
Mixture with citric acid.Nomenclature
Description and references
Clinical trial in treatment of apnea in premature neonates: P. B. Larsen et al., Acta Paediatr. 84, 360 (1995).
Properties
White, crystalline powder; acid reaction. Sol in about 4 parts warm water.Therapeutic Category
CNS stimulant; respiratory stimulant.
Therapeutic Category (Veterinary)
Cardiac and respiratory stimulant; diuretic.Keywords
CNS Stimulant