1750. Canrenone
Nomenclature
CAS number: 976-71-6
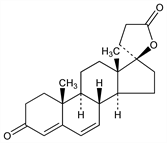
(17α)-17-Hydroxy-3-oxopregna-4,6-diene-21-carboxylic acid γ-lactone; 17α-(2-carboxyethyl)-17β-hydroxyandrosta-4,6-dien-3-one lactone; 17α-(2-carboxyethyl)-17β-hydroxy-3-oxoandrosta-4,6-diene lactone; 6-dehydrotestosterone-17α-propionic acid γ-lactone; 3-(3-oxo-17β-hydroxy-4,6-androstadien-17α-yl)propionic acid γ-lactone; Phanurane (Specia). C22H28O3; mol wt 340.46.
C 77.61%, H 8.29%, O 14.10%.
Description and references
Aldosterone antagonist. Prepd by dehydrogenation of 17-hydroxy-3-oxo-17α-pregn-4-ene-21-carboxylic acid γ-lactone: Cella, Tweit, J. Org. Chem. 24, 1109 (1959); Cella, US 2900383 (1959 to Searle).
Properties
Crystals from ethyl acetate, mp 149-151°, solidifies and remelts at 165°. [α]D +24.5° (chloroform). uv max: 283 nm (ε 26700).Derivative
Free acid potassium salt.Nomenclature
CAS number: 2181-04-6
Potassium canrenoate; Kanrenol (SPA); Soldactone (Searle); Venactone (Lepetit). C22H29KO4; mol wt 396.56.
C 66.63%, H 7.37%, K 9.86%, O 16.14%.
Therapeutic Category
Diuretic.
Keywords
Aldosterone Antagonist; Diuretic; Steroids