1776. Caramiphen
Nomenclature
CAS number: 77-22-5
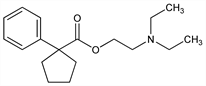
1-Phenylcyclopentanecarboxylic acid 2-(diethylamino)ethyl ester; diethylaminoethyl 1-phenylcyclopentane-1-carboxylate. C18H27NO2; mol wt 289.41.
C 74.70%, H 9.40%, N 4.84%, O 11.06%.
Description and references
Anticholinergic. Prepn: CH 234452; H. Martin, F. H"afliger, US 2404588 (1945, 1946 both to Geigy). Prepn of the ethanedisulfonate: CH 272708 (1951 to Geigy), C.A. 46, 4563i (1952); Chem. Zentralbl. 1952, 1571. Pharmacology and toxicology of the hydrochloride: C. P. Kraatz et al., J. Pharmacol. Exp. Ther. 96, 42 (1949); of the ethanedisulfonate: J. J. Toner, E. Macko, ibid. 106, 246 (1952). GLC determn in blood: P. Levandoski, T. Flanagan, J. Pharm. Sci. 69, 1353 (1980). Mechanism of antitussive action: E. F. Domino et al., J. Pharmacol. Exp. Ther. 233, 249 (1985).
Properties
bp0.07 112-115°.Derivative
Ethanedisulfonate.Nomenclature
CAS number: 125-86-0
(C18H27NO2)2.C2H6O6S2; mol wt 769.02.
C 59.35%, H 7.86%, N 3.64%, O 20.80%, S 8.34%.
Properties
Crystals from acetone, mp 115-116°. More sol in water than the hydrochloride. Sol in alc, pharmaceutical syrups. Mixture with phenylpropanolamine hydrochloride, Tuss-Ornade.Derivative
Hydrochloride.Nomenclature
CAS number: 125-85-9
C18H27NO2.HCl; mol wt 325.87.
C 66.34%, H 8.66%, N 4.30%, O 9.82%, Cl 10.88%.
Properties
Crystals, mp 145-146°. Sol in alc; slightly sol in water. LD50 i.p. in rats: 209 mg/kg (Kraatz).Therapeutic Category
Antitussive.
Keywords
Antitussive