1792. Carbenicillin
Nomenclature
Description and references
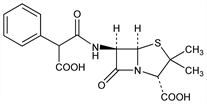
Semi-synthetic antibiotic related to penicillin. Prepn of monopotassium salt: Hobbs, US 3142673 (1964 to Pfizer); of disodium salt: BE 646991; Brain, Nayler, US 3282926 (1964, 1966 both to Beecham). Activity studies and pharmacology: Naumann, Kempf, Arzneim.-Forsch. 19, 1222 (1969). Chemistry and mode of action: Butler et al., J. Infect. Dis. 122, Suppl., 81 (1970). Clinical data: Hoffler et al., ibid. 1233; Gritz, Naumann, ibid. 1237. HPLC determn of epimers in plasma and urine: M. Ishida et al., J. Chromatogr. B 652, 43 (1994). Review of antibacterial activity, pharmacology, and clinical use: H. C. Neu, Med. Clin. North Am. 66, 61-76 (1982).
Derivative
Disodium salt.Nomenclature
Properties
White powder. LD50 i.p. in rats: >2000 mg/kg See: E. I. Goldenthal, Toxicol. Appl. Pharmacol. 18, 185 (1971).Derivative
Phenyl sodium.Nomenclature
Description and references
Prepn and activity studies: Clayton et al., J. Med. Chem. 18, 172 (1975).
Properties
Crystals from ethanol, [α]D20 +216.2° (H2O).Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antimicrobial in reptiles.Keywords
Antibacterial (Antibiotics); β-Lactams; Penicillins