1797. Carbidopa
Nomenclature
Description and references
Peripheral decarboxylase inhibitor. Prepn of dl-form: Pfister, FR M1553 (1962 to Merck & Co.), C.A. 59, 12921e (1963); Sletzinger et al., J. Med. Chem. 6, 101 (1963); GB 940596; Chemerda et al., US 3462536 (1963, 1969 both to Merck & Co.). Synthesis of the l-form: Karady et al., DE 2062285; DE 2062332 (both 1971 to Merck & Co.), C.A. 75, 118122t, 118120r (1971); eidem, J. Org. Chem. 36, 1946, 1949 (1971). Inhibition of dopa decarboxylase: Porter et al., Biochem. Pharmacol. 11, 1067 (1962); Moran, Sourkes, J. Pharmacol. Exp. Ther. 148, 252 (1962); Watanabe et al., Clin. Pharmacol. Ther. 11, 740 (1970). Only the l-form is pharmacologically active: Lotti, Porter, J. Pharmacol. Exp. Ther. 172, 406 (1970).
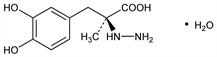
Properties
Crystals from hot water, mp 203-205° (dec). [α]D -17.3° (methanol). Also reported as mp 208°.Derivative
Combination with levodopa.Nomenclature
Derivative
dl-Form.Properties
Tan fluffy crystals, mp 206-208° (dec). uv max (methanol): 282.5 nm (ε 2940).Therapeutic Category
In combination with levodopa as antiparkinsonian.
Keywords
Antiparkinsonian