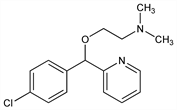
1799. Carbinoxamine
Nomenclature
CAS number: 486-16-8
2-[(4-Chlorophenyl)-2-pyridinylmethoxy]-N,N-dimethylethanamine; 2-[p-chloro-α-(2-dimethylaminoethoxy)benzyl]pyridine; paracarbinoxamine. C16H19ClN2O; mol wt 290.79.
C 66.09%, H 6.59%, Cl 12.19%, N 9.63%, O 5.50%.
Description and references
Prepn: Tilford, Shelton, US 2606195 (1952 to Wm. S. Merrell); Swain, US 2800485 (1957 to McNeil Labs.). Prepn of l-form: GB 905993 (1962 to McNeil Labs.), C.A. 58, 5644a (1962). Abs config of l-form: V. Barouh et al., J. Med. Chem. 14, 834 (1971). Pharmacology and toxicology: R. Cahen, Ann. Pharm. Fr. 20, 463 (1962). GLC determn in serum: D. J. Hoffman et al., J. Pharm. Sci. 72, 1342 (1983).
Properties
Liquid, bp0.1 158-162°.Derivative
Hydrochloride.C16H19ClN2O.HCl; mol wt 327.25.
C 58.72%, H 6.16%, Cl 21.67%, N 8.56%, O 4.89%.
Properties
Crystals from isopropanol + ethyl acetate, dec 162-164°. Sol in water.Derivative
Maleate.Nomenclature
CAS number: 3505-38-2
Allergefon (Lafon); Clistin (McNeil); Ciberon (Taisho); Lergefin (Larma); Polistin T-Caps (Trommsdorff). C16H19ClN2O.C4H4O4; mol wt 406.86.
C 59.04%, H 5.70%, Cl 8.71%, N 6.89%, O 19.66%.
Properties
Bitter crystals from ethyl acetate, mp 117-119°. Freely sol in water, alcohol, chloroform. Very slightly sol in ether. pH of 1% aq soln 4.6-5.1. LD50 in mice (mg/kg): 166 i.p. (Cahen).Derivative
l-Form.Nomenclature
Levocarbinoxamine; rotoxamine; McN-R-73-Z.Properties
bp0.5 143-144°. nD20 1.5522. [α]D25 -6.8° (c = 2 in methanol).Derivative
l-Form d-tartrate.Nomenclature
CAS number: 49746-00-1
Twiston (McNeil). C16H19ClN2O.C4H6O6; mol wt 440.87.
C 54.49%, H 5.72%, Cl 8.04%, N 6.35%, O 25.40%.
Properties
Crystals from isopropanol, mp 143-144.5°. [α]D25 +37.2° (c = 20 in methanol).Therapeutic Category
Antihistaminic.
Keywords
Antihistaminic; Aminoalkylethers