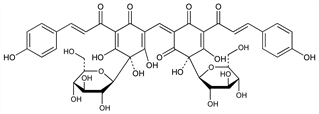
1867. Carthamin
Nomenclature
Description and references
Coloring principle from safflower, Carthamus tinctorius L., Compositae. Purification and review of early efforts: T. Kametaka, A. G. Perkin, J. Chem. Soc. 97, 1415 (1910). Structure: C. Kuroda, J. Chem. Soc. 1930, 752, 765. Revised structure: H. Obara, J. Onodera, Chem. Lett. 1979, 201. Absolute configuration: S. Sato et al., ibid. 1996, 833. Total synthesis as acetate: S. Sato et al., ibid. 2001, 1318. Biogenesis: M. Shimokoriyama, S. Hattori, Arch. Biochem. Biophys. 54, 93 (1955). Determn of levels in Carthamus Red: T. Morimoto et al., Nippon Shokuhin Kagaku Gakkaishi 5, 236 (1998). Color stability in aqueous soln: J.-B. Kim, Y.-S. Paik, Arch. Pharmacal Res. 20, 643 (1997); with polysaccharides: K. Saito, Acta Bot. Croat. 57, 123 (1998). Review of prepn protocols: K. Saito, Y. Fukaya, Acta Alimentaria 26, 141-152 (1997). See also: Colour Index vol. 4 (3rd ed., 1971) p 4625.