1891. Cassaine
Nomenclature
CAS number: 468-76-8
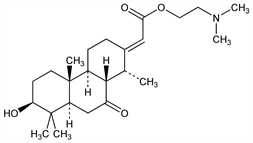
[1R-(1α,2E,4aα,4bβ,7β,8aα,10aβ)]-(Dodecahydro-7-hydroxy-1,4b,8,8-tetramethyl-10-oxo-2(1H)-phenanthrenylidene)acetic acid 2-(dimethylamino)ethyl ester; (E)-3β-hydroxy-14α-methyl-7-oxopodocarpane-Δ13,α-acetic acid 2-(dimethylamino)ethyl ester. C24H39NO4; mol wt 405.57.
C 71.07%, H 9.69%, N 3.45%, O 15.78%.
Description and references
Cardiotonic principle from the bark of Erythrophleum guineense G. Don., Leguminosae: Dalma, Helv. Chim. Acta 22, 1497 (1939). Structure: Turner et al., Tetrahedron Lett. 1959, 7; Gensler et al., J. Am. Chem. Soc. 81, 5217 (1959). Absolute configuration: Hauth et al., Helv. Chim. Acta 48, 1087 (1965); Clarke et al., J. Am. Chem. Soc. 88, 5865 (1966). Synthesis: Turner et al., ibid. 1766.
Properties
Glossy flakes from ether, mp 142.5°. [α]D23 -110.5° (alc); [α]D23 -101° (0.1N HCl). Absorption spectrum: Ruzicka, Dalma, Helv. Chim. Acta 22, 1516 (1939). Sol in methanol, ethanol, acetone, acetic acid, chloroform, ether, benzene.Derivative
Hydrochloride hydrate.C24H40ClNO4.H2O; mol wt 460.05.
C 62.66%, H 9.20%, Cl 7.71%, N 3.04%, O 17.39%.