1917. Cefazolin
Nomenclature
Description and references
Semi-synthetic antibiotic derived from 7-aminocephalosporanic acid, q.v. Prepn: T. Takano et al., ZA 6804513; eidem, US 3516997 (1969, 1970 to Fujisawa). Synthesis and properties: Kariyone et al., J. Antibiot. 23, 131 (1970). Activity and clinical studies: Nishida et al., ibid. 137, 184; Shibata, Fujii, Antimicrob. Agents Chemother. 1970, 467. HPLC determn in plasma and tissue: S. M. Bayoumi et al., Int. J. Pharm. 30, 57 (1986); in ophthalmic ointment: C. F. Martin et al., J. Chromatogr. 402, 376 (1987). Antibacterial activity of crystalline modifications: G. Opalchenova, G. N. Kalinkova, Int. J. Pharm. 189, 235 (1999). Metabolic studies: Kozatani et al., Chem. Pharm. Bull. 20, 1105 (1972). Toxicology: H. A. Birkhead et al., J. Infect. Dis. 128, Suppl., 379 (1973). Comprehensive description: A. E. Zappala et al., Anal. Profiles Drug Subs. 4, 1-20 (1975). Review: H. Nakano in Pharmacological and Biochemical Properties of Drug Substances vol. 1, M. E. Goldberg, Ed. (Am. Pharm. Assoc., Washington, DC, 1977) pp 155-182.
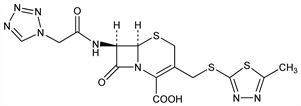
Properties
Needles from aq acetone, mp 198-200° (dec). uv max (buffer pH 6.4): 272 nm (ε 13150). Easily sol in DMF, pyridine; sol in aq acetone, aq dioxane, aq ethanol; slightly sol in methanol. Practically insol in chloroform, benzene, ether.Derivative
Sodium salt.Nomenclature
Properties
White to yellowish-white, odorless crystalline powder with a bitter, salty taste. Crystallizes in α, β, and γ-forms (Kariyone). Easily sol in water, slightly sol in methanol, ethanol. Practically insol in benzene, acetone, chloroform. LD50 in mice, rats (mg/kg): 3.9, 3.18 i.v.; 6.2, 7.4 i.p. (Birkhead).Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Antibiotics); β-Lactams; Cephalosporins