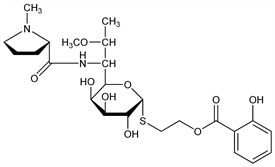
Nomenclature
CAS number: 2520-21-0
2-[(2-Hydroxybenzoyl)oxy]ethyl 6,8-dideoxy-7-
O-methyl-6-[[[(2
S)-1-methyl-2-pyrrolidinyl]carbonyl]amino]-1-thio-
d-
erythro-α-
d-
galacto-octopyranoside; α-2-hydroxyethyl 6,8-dideoxy-7-
O-methyl-6-(1-methyl-
l-2-pyrrolidinecarboxamido)-1-thio-
d-
erythro-d-
galacto-octopyranoside monosalicylate;
S-demethyl-3′-depropyl-
S-[2-[(2-hydroxybenzoyl)oxy]ethyl]-7-
O-methyllincomycin.
C
24H
36N
2O
9S; mol wt 528.62.
C 54.53%, H 6.86%, N 5.30%, O 27.24%, S 6.07%.
Description and references
Antibiotic substance produced by Streptomyces
caelestis from a soil obtained from Emigration Canyon near Salt
Lake City: DeBoer et al., Antibiot.
Annu. 1954-1955, 831; Hoeksema et al., ibid. 837. Structure: Hoeksema, J. Am. Chem. Soc. 86, 4224 (1964); 90, 755 (1968).
Properties
Amphoteric, amorphous base. [α]D24 +126.6° (c = 0.5 in chloroform). uv max (0.01N alcoholic H2SO4): 240, 310 nm (E1%1cm 183.7, 80.6). Soluble
in acidic or strongly basic aq solns, practically insol in the pH
range of 7 to 10. Soluble in the more polar organic solvents, but
not in ether or light hydrocarbons. Active against gram-positive
organisms and plant pathogens such as B. stewartii, P. fascians,
X. pruni at 0.5 γ/ml. The antibacterial activity is rapidly
destroyed in aq solns above pH 9 at 24°, but remains at pH 5 to 7
for at least 60 days. Celesticetin is more stable in acid than in
basic solns, but is destroyed rapidly in 1N HCl at 100°.
Cross resistance with erythromycin.Derivative
Salicylate.
Properties
Monoclinic tablets, mp 139°. [α]D24 +90.2° (c = 0.5 in H2O). Sol in water.Derivative
Oxalate.
Properties
Crystals, mp 149-154°. [α]D24 +106.6° (c = 0.5 in H2O). Sol in water.