1982. Cephalothin
Nomenclature
CAS number: 153-61-7
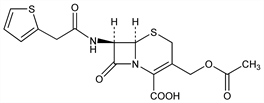
(6R,7R)-3-[(Acetyloxy)methyl]-8-oxo-7-[(2-thienylacetyl)amino]-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic
acid; 3-(hydroxymethyl)-8-oxo-7-[2-(2-thienyl)acetamido]-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic
acid acetate; 7-(2-thienylacetamido)cephalosporanic acid; 7-(thiophene-2-acetamido)cephalosporanic acid. C16H16N2O6S2; mol wt 396.44.
C 48.47%, H 4.07%, N 7.07%, O 24.21%, S 16.18%.
Description and references
Semi-synthetic cephalosporin antibiotic. Prepn: Chauvette et al., J. Am. Chem. Soc. 84, 3401 (1962); E. H. Flynn, US 3218318 (1965 to Lilly); FR 1384197 (1965 to Glaxo), C.A. 63, 11591c (1965). Bacteriology and pharmacology: Lee, Anderson, Antimicrob. Agents Chemother. 1962, 695; Walters et al., ibid. 706; Naumann, Arzneim.-Forsch. 16, 1099 (1966). Acute toxicity: M. Kuramoto et al., Jpn. J. Antibiot. 27, 746 (1974), C.A. 83, 71972t (1975). Comprehensive description: R. J. Simmons, Anal. Profiles Drug Subs. 1, 319-341 (1972).
Properties
mp 160-160.5°. [α]D20 +50° (c = 1.03 in acetonitrile), see R. B. Woodward et al., J. Am. Chem. Soc. 88, 852 (1966).Derivative
Sodium salt.Nomenclature
CAS number: 58-71-9
Averon-1 (Alfar); Cefalotin; Cemastin (Ist. Vitamine); Cephation (Meiji); Ceporacin (Torii); Cepovenin (Hoechst; Glaxo); Chephalotin (Lilly); Coaxin (Tobishi); Keflin (Lilly); Lospoven (Hoechst); Synclotin (Toyo Jozo); Toricelocin (Torii). C16H15N2NaO6S2; mol wt 418.42.
C 45.93%, H 3.61%, N 6.70%, Na 5.49%, O 22.94%, S 15.33%.
Properties
mp 204-205°. [α]D +135° (c = 1.0 in water). uv max: 236, 260 nm (ε 12950, 9350). LD50 in mice, rats (mg/kg): >20000, >10000 orally; 5670, 7716 i.p. (Kuramoto).Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Antibiotics); β-Lactams; Cephalosporins