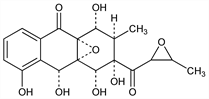
2007. Cervicarcin
Nomenclature
CAS number: 18700-78-2
1,2,3,4-Tetrahydro-1,3,4,5,10-pentahydroxy-2-methyl-3-[(3-methyloxiranyl)carbonyl]-4a,9a-epoxyanthracen-9(10H)-one; 4a,9a-epoxy-3-(2,3-epoxybutyryl)-1,2,3,4,4a,9a-hexahydro-1,3,4,5,10-pentahydroxy-2-methylanthrone. C19H20O9; mol wt 392.36.
C 58.16%, H 5.14%, O 36.70%.
Description and references
Antineoplastic antibiotic produced by Streptomyces ogaensis: Okuma et al., J. Antibiot. 15A, 152, 247 (1962). Prepn: Sumiki et al., JP 64 7400 (1964 to Inst. Phys. & Chem. Res.). Structure: Marumo et al., J. Am. Chem. Soc. 86, 4507 (1964); eidem, Agric. Biol. Chem. 32, 209 (1968). Stereochemistry: eidem, ibid. 35, 1931 (1971). Activity studies: C. Itakura et al., J. Antibiot. 16A, 231 (1963).