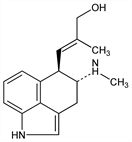
2042. Chanoclavine
Nomenclature
Description and references
Precursor of the tetracyclic ergolines, agroclavine, elymoclavine, and lysergic acid amide, q.q.v.: Gr"oger et al., Z. Naturforsch. 21b, 827 (1966); Floss et al., Chem. Commun. 1967, 105. Occurs in sclerotia of Claviceps purpurea (Fries) Tul., Hypocreaceae, in Ipomea tricolor Cav., Convolvulaceae and is one of the active principles of the ancient Aztec drug “Ololiuqui,” Rivea coryrubosa (L.) Hall. f., Convolvulaceae: Hofmann, Tscherter, Experientia 16, 414 (1960). Isoln and structure: Hofmann et al., Helv. Chim. Acta 40, 1358 (1957). Isoln of stereoisomers: Stauffacher, Tscherter, ibid. 47, 2186 (1964). Stereochemistry: Acklin et al., Chem. Commun. 1966, 799. Biosynthetic studies: Floss et al., J. Am. Chem. Soc. 90, 6500 (1968). Total synthesis of (±)-chanoclavine: H. Plieninger et al., Tetrahedron Lett. 1975, 1827; H. Plieninger, D. Schmalz, Ber. 109, 2140 (1976); M. Natsume, H. Muratake, Heterocycles 16, 375 (1981).