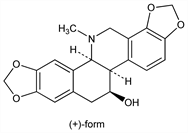
2053. Chelidonine
Nomenclature
Stylophorin.Description and references
Hexahydrobenzophenanthridine alkaloid. Occurs in nature in (+)-form, (-)-form and racemic form. Isoln of (+)-form from root of Chelidonium majus L., Stylophorum diphyllum (Michx.) Nutt., and Dicranostigma franchetianum (Prain) Fedde, Papaveraceae: J. M. Probst, Ann. 29, 113 (1839); E. Schmidt, F. Selle, Arch. Pharm. 228, 441 (1890); F. Selle, ibid. 96; Manske, Can. J. Res. 20B, 53 (1942); J. Slavik, Collect. Czech. Chem. Commun. 20, 198 (1955); from Symphoricarpos albus L., Blake, Caprifoliaceae: M. Szaufer et al., Phytochemistry 17, 1446 (1978). Isoln of (-)-form from Glaucium corniculatum Curt., Papaveraceae: J. Slavik, L. Slavikova, Collect. Czech. Chem. Commun. 22, 279 (1957). Structure: F. von Bruchhausen, H. W. Bersch, Ber. 63, 2520 (1930); E. Sp"ath, F. Kuffner, ibid. 64, 370 (1931); H. W. Bersch, Arch. Pharm. 291, 491 (1958). Identity of (±)-form with diphylline: J. Slavik, Collect. Czech. Chem. Commun. 26, 2933 (1961). Absolute configuration of (+)-p-bromobenzoate: N. Takao et al., Tetrahedron Lett. 1979, 495. Conformation: M. Cushman, T.-C. Choong, Heterocycles 14, 1935 (1980); M. Sugiura et al., J. Chem. Soc. Perkin Trans. 2 1986, 175. Chiroptic properties and absolute configuration of (+)-chelidonine: N. Takao et al., Arch. Pharm. 317, 223 (1984). Biosynthesis: E. Leete, J. Am. Chem. Soc. 85, 473 (1963). Total synthesis of (±)-form: W. Oppolzer, K. Keller, ibid. 93, 3836 (1971); M. Cushman et al., J. Org. Chem. 45, 5067 (1980); W. Oppolzer, C. Rabbiani, Helv. Chim. Acta 66, 1119 (1983); M. Hanaoka et al., Chem. Lett. 1986, 736. Effect on smooth muscle: P. J. Hanzlik: J. Pharmacol. Exp. Ther. 7, 99 (1915). Inhibition of reverse transcriptase activity: M. L. Sethi, Can. J. Pharm. Sci. 16, 29 (1981). Cytotoxic effects: M. Cushman et al., loc. cit. Review of pharmacological effects: V. Preininger, “The Biology of Papaveraceae Alkaloids” in The Alkaloids vol. 15, R. H. F. Manske, Ed. (Academic Press, Orlando, 1975) pp 241-242. General review: V. Simanek, “Benzophenanthridine Alkaloids” ibid. vol. 26, A. Brossi, Ed. (1985) pp 185-240. Toxicity data: R. C. Anderson, K. K. Chen, Fed. Proc. 5, 163 (1946).