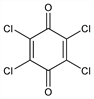
2078. Chloranil
Nomenclature
CAS number: 118-75-2
2,3,5,6-Tetrachloro-2,5-cyclohexadiene-1,4-dione; 2,3,5,6-tetrachloro-p-benzoquinone; tetrachloroquinone; Spergon; Vulklor. C6Cl4O2; mol wt 245.88.
C 29.31%, Cl 57.68%, O 13.01%.
Description and references
Prepd from p-phenylenediamine or phenol by treating with KClO3 and HCl. Because of its great resistance to further oxidation, chloranil is formed as the final product of the chlorate-HCl oxidation of many aromatic compds. For comprehensive list see Huntress, Organic Chlorine Compounds (New York, 1948). Laboratory procedure starting with phenol or p-chlorophenol: Fierz-David, Blangey, Grundlegende Operationen der Farbenchemie (Vienna, 5th ed., 1943) p 140. Use as reagent for pamaquine, q.v., in urine: Schulemann et al., Chem. Zentralbl. 1928, I, 2193. Aqueous decomposition study: D. H. Sarr et al., Environ. Sci. Technol. 29, 2735 (1995). Review: Health and Environmental Effects Profile for Chloranil (EPA PB88-219696, 1986) 59 pp.