Nomenclature
CAS number: 78-95-5
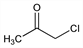
1-Chloro-2-propanone; chloracetone; monochloroacetone; monochloracetone; acetonyl chloride; chloropropanone; 1-chloro-2-ketopropane; 1-chloro-2-oxopropane.
C
3H
5ClO; mol wt 92.52.
C 38.95%, H 5.45%, Cl 38.32%, O 17.29%.
Description and references
Prepn by the action of chlorine upon diketene:
A. B. Boese, Jr., US 2209683 (1940 to Carbide and Carbon Chem.); by chlorination
of acetone: E. J. Rahrs, US 2235562 (1941 to Eastman Kodak); G. H. Morey, US 2243484 (1941 to Commercial
Solvents Corp.). Stabilization: idem, US 2229651 (1941 to Commercial Solvents Corp.); E. J. Rahrs, US 2263010 (1941 to Eastman Kodak). Forms binary azeotropes with many organic liquids: L.
H. Horsley, Azeotropic Data, Advances in Chemistry Series
No. 6 (Washington, 1952) p 73. Reacts with aryl Grignard reagents
to form stilbenes: Huang, J. Chem. Soc. 1954, 2539. Toxicology: E. V. Sargent et al., Am. Ind. Hyg. Assoc. J. 47, 375 (1986).
Properties
Liquid. Pungent odor. Lacrimator. Turns dark
and resinifies on prolonged exposure to light. May be stabilized
by addition of 0.1% H2O or 1.0% CaCO3. d425 1.123; d415 1.135. mp -44.5°. bp 119.7°, bp50 61°, bp12 20°. Volatile with steam. Surface tension at 20° = 35.27 dyn/cm.
Poisonous, flammable, corrosive. Dipole moment in hexane, 2.36D. Sol in 10 parts water (w/w).
Miscible with alcohol, ether, chloroform. LD50 (14 day) in mice, rats (mg/kg):
127, 100 orally; LC50 (1 hr) in rats (ppm): 262 by inhalation (Sargent).Derivative
2,4-Dinitrophenylhydrazone.
Properties
Yellow needles from alc, mp 124°.Caution
Intensely irritating to eyes, skin,
mucous membranes.Use
Has been proposed as tear gas component for police
and military use; in the manuf of couplers for color photography;
as enzyme inactivator; intermediate in the manuf of perfumes, antioxidants,
drugs; in insecticide formulations; in photopolymerization of vinyl
compds. Proposed as catalyst in tetraethyllead production, as selective
solvent for separating diolefins.
