2118. Chloroaniline
C6H6ClN; mol wt 127.57.
C 56.49%, H 4.74%, Cl 27.79%, N 10.98%.
Description and references
Prepn of m-, o- and p-isomers: Sidgwick, Rubie, J. Chem. Soc. 119, 1013 (1921). Manufacture of m- and p-isomers by catalytic hydrogenation of chloronitrobenzene: Pray, Trager, US 2791613 (1957 to Columbia-Southern Chem.); by reduction of chloronitrobenzene with NaHS: Latourette et al., US 2894035 (1959 to Food Machinery & Chem.). Properties of o-isomer: Dreisbach, Martin, Ind. Eng. Chem. 41, 2875 (1949) DOI. Toxicity data: H. F. Smyth et al., Am. Ind. Hyg. Assoc. J. 23, 95 (1962) PubMed.
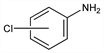
Derivative
m-Chloroaniline.Nomenclature
CAS number: 108-42-9
3-Chlorobenzenamine. Properties
Liquid, bp 230.5°. mp -10.4°. d422 1.2150. nD20 1.5931. Poisonous. Practically insol in water. Sol in most common organic solvents.Derivative
o-Chloroaniline.Nomenclature
CAS number: 95-51-2
Properties
Liquid. At 99.61 mol-% purity, bp 208.84°; mp -1.94°. d422 1.2114. nD20 1.5895. Poisonous. Practically insol in water. Sol in acid and in most organic solvents.Derivative
p-Chloroaniline.Nomenclature
CAS number: 106-47-8