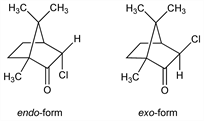
2132. 3-Chloro-d-camphor
Nomenclature
CAS number: 508-29-2
3-Chloro-1,7,7-trimethylbicyclo[2.2.1]heptan-2-one; 3-chloro-d-2-bornanone. C10H15ClO; mol wt 186.68.
C 64.34%, H 8.10%, Cl 18.99%, O 8.57%.
Description and references
Of the two isomers, the endo-form is more stable than the exo-form: Cookson, J. Chem. Soc. 1954, 282. Prepn of endo-form: Kipping, Pope, ibid. 63, 548 (1893). Prepn of exo-form by isomerization of endo-form: Lowry, Steele, ibid. 107, 1382 (1915). uv spectra: Lowry, Owen, J. Chem. Soc. 129, 606 (1926). Crystal structure of endo-form: Wiebenga, Krom, Rec. Trav. Chim. 65, 663 (1946). Configuration: Cookson, loc. cit.; Kumler et al., J. Am. Chem. Soc. 83, 2711 (1961). Review: The Terpenes vol. II, J. L. Simonsen, Ed. (University Press, Cambridge, 2nd ed., 1949) p 397.