2151. Chloronitrobenzene
Nomenclature
Nitrochlorobenzene.C6H4ClNO2; mol wt 157.55.
C 45.74%, H 2.56%, Cl 22.50%, N 8.89%, O 20.31%.
Description and references
Prepn of m-isomer: W. W. Hartman, M. R. Brethen, Org. Synth. coll. vol. I (2nd ed., 1964) p 162.
Derivative
m-Chloronitrobenzene.Nomenclature
CAS number: 121-73-3
1-Chloro-3-nitrobenzene; m-nitrochlorobenzene. Properties
Pale-yellow orthorhombic prisms from alcohol. d420 1.534. mp 46°. bp760 236°; bp12 117°. Poisonous. Insol in water. Sparingly sol in cold alcohol; freely sol in hot alcohol, chloroform, ether, carbon disulfide, glacial acetic acid.Derivative
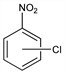
o-Chloronitrobenzene.Nomenclature
CAS number: 88-73-3
Properties
Yellow crystals. d 1.305. mp 32-33°. bp 245-246°. Poisonous. Insol in water. Sol in alcohol, benzene, ether.Derivative
p-Chloronitrobenzene.Nomenclature
CAS number: 100-00-5