2155. Chlorophyll
Nomenclature
Description and references
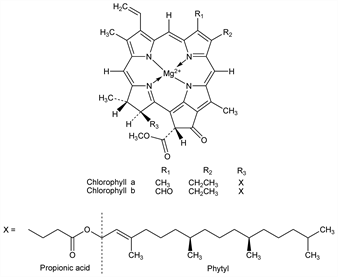
The green, photosynthetic pigment of plants. Higher plants and green algae contain chlorophyll a and chlorophyll b in the approx ratio of 3:1. Chlorophyll c is found together with chlorophyll a in many types of marine algae: Jeffrey, Biochem. J. 86, 313 (1963). Red algae (Rhodophyta) contain principally chlorophyll a and also chlorophyll d: Manning, Strain, J. Biol. Chem. 151, 1 (1943). Isoln by chromatography: Tswett, Ber. Dtsch. Bot. Ges. 24, 316, 385 (1906); Willst"atter, Stoll, Investigations on Chlorophyll (transl by Schertz and Merz: Lancaster, 1928); Schertz, Ind. Eng. Chem. 30, 1073 (1938); Fischer-Orth-Stern, Die Chemie des Pyrrols Vol. II, part 2 (Leipzig, 1940); Zechmeister, Cholnoky, Principles and Practice of Chromatography (New York, 1943). Industrial large-scale isoln processes: Judah et al., Ind. Eng. Chem. 46, 2262 (1954). Structure: Fischer-Orth-Stern, loc. cit.; Ficken et al., J. Chem. Soc. 1956, 2273. Total synthesis of chlorophyll a: R. B. Woodward et al., J. Am. Chem. Soc. 82, 3800 (1960); idem, Angew. Chem. 72, 651 (1960); Strell et al., ibid. 169; R. B. Woodward, Pure Appl. Chem. 2, 383 (1963); idem et al., Tetrahedron 46, 7599 (1990). Abs config of chlorophyll a: I. Fleming, Nature 216, 151 (1967); of chlorophylls a and b: Brockmann, Ann. 754, 139 (1971); Brockmann, Bode, Ann. 1974 (7), 1017. 13C-NMR study of chlorophyll a: S. L"otj"onen, P. H. Hynninen, Org. Magn. Reson. 16, 304 (1981); of chlorophyll b: N. Risch, H. Brockmann, Tetrahedron Lett. 24, 173 (1983). Review of syntheses: Johnson, Sci. Prog. 49, 77 (1961). Comprehensive reviews with bibliography: Stoll, Wiedemann, Fortschr. Chem. Org. Naturst. 1, 159-254 (1938); Fortschr. Chem. Forsch. 2, 538 (1952); The Chlorophylls, L. P. Vernon, G. R. Seely, Eds. (Academic Press, New York, 1966) 679 pp; Inhoffen et al., Fortschr. Chem. Org. Naturst. 26, 284-298 (1968); Inhoffen, Pure Appl. Chem. 17, 443-460 (1968).
Derivative
Chlorophyll a.Nomenclature
Properties
Sepn and purification: Anderson, Calvin, Nature 194, 285 (1962). Waxy blue-black microcrystals, usually aggregates of thin, lancet-like leaflets, mp 117-120°. [α]D20 -262° (acetone). Absorption max (ether): 660, 613, 577, 531, 498, 429, 409 nm. Freely sol in ether, ethanol, acetone, chloroform, carbon disulfide, benzene. Sparingly sol in cold methanol. Practically insol in petr ether. The alcoholic soln is blue-green with a deep-red fluorescence.Derivative
Chlorophyll b.Nomenclature
Properties
Waxy blue-black microcrystals. Sinters between 86° and 92°, becomes a viscous liquid at 120-130° and then begins to puff up in large bubbles. [α]D20 -267° (acetone-methanol). Absorption max (ether): 642, 593, 565, 545, 453, 427 nm. Sparingly sol in petr ether, ligroin, cold methanol. Freely sol in abs alcohol, ether. The ether soln has a brilliant green color. Solns with other organic solvents are usually green to yellowish-green with red fluorescence.Derivative
Chlorophyll c.Properties
Structure: Dougherty et al., J. Am. Chem. Soc. 88, 5037 (1966). Reddish-black hexagonal bipyramids or four-sided plates from dil ethanol. Absorption max (acetone): 628, 580, 442 nm (E1cm0.1% 15.8, 10.7, 115.9). Sol in methanol, ethanol, ethyl acetate; practically insol in ether, acetone.Derivative
Chlorophyll d.Properties
Chlorophyll a with the 9-vinyl group replaced by a formyl group: Holt, Morley, Can. J. Chem. 37, 507 (1959); Holt, Can. J. Bot. 39, 327 (1961). Absorption max (ether): 686, 445 nm. The chlorophyll of commerce is an intensely dark-green, aq, alc, or oil soln. Careful alkaline hydrolysis of chlorophyll opens the cyclopentanone ring and replaces the methyl and phytyl ester groups with Na or K; the resulting salts are called chlorophyllins and are water sol, e.g. sodium magnesium chlorophyllin, C34H31N4Na3MgO6. Acid treatment of chlorophyll removes the Mg replacing it with H2 which can be replaced with other metals, e.g. iron pheophytin, C55H72FeN4O5, sol in oil. Comprehensive review: “Chlorophyll Derivatives, Their Chemistry, Commercial Preparation and Uses” by J. C. Kephart in Econ. Bot. 9, 3-38 (1955). Review of commercial chlorophyllin prepns: Strell et al., Arzneim.-Forsch. 5, 640 (1955); 6, 8 (1956). Trademarks: Chloresium, Chlorofolin, Darotol, Ennds, Exodor-Grun, Nullo Chlorophyll, Stozzon-Chlorophyll.Use
To color soaps, oils, fats, waxes, confectionery, preserves, liquors, cosmetics, perfumes. Source of phytol. For dyeing leather. As sensitizer for color film. Has been used as antiknock agent in gasoline; as accelerator in the vulcanizing of rubber; in deodorizers.Therapeutic Category
Deodorant.