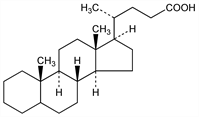
2196. Cholanic Acid
Nomenclature
Description and references
Steroidal acid probably formed by the dehydration and hydrogenation of certain bile acids commonly found in animals. Considered to be a chemical trademark certifying the prehistoric presence of some type of animal, see Seifert, Pure Appl. Chem. 34, 633 (1973). This status as a natural product of exclusive animal origins now questioned by its isolation from the embryo of the jequirity bean, Abrus precatorius, Leguminosae: Mandava et al., Steroids 23, 357 (1974). Prepn from coprostane, cholic acid, lithocholic acid, desoxy- and chenodesoxycholic acid: Wieland, Weil, Z. Physiol. Chem. 80, 287 (1912); Wieland, Boersch, ibid. 106, 193 (1919); Windaus, Neukirchen, Ber. 52, 1915 (1919); Wieland, Vocke, Z. Physiol. Chem. 191, 69 (1930). From 3-ketocholanic acid diethyl thioacetal by hydrogenolysis: Bernstein, Dorfman, US 2440660 (1948 to Am. Cyanamid). 5β-Cholanic acid differs from the thermodynamically more stable 5α-isomer, by being cis at the A/B steroid ring function rather than trans.