2212. Chondrocurine
Nomenclature
CAS number: 477-58-7
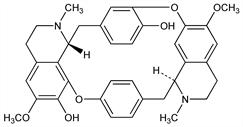
6,6′-Dimethoxy-2,2′-dimethyltubocuraran-7′,12′-diol; d-chondocurine; d-tubocurine. C36H38N2O6; mol wt 594.70.
C 72.71%, H 6.44%, N 4.71%, O 16.14%.
Description and references
Isoln from Chondodendron tomentosum Ruiz & Pav., Menispermaceae: Wintersteiner, Dutcher, Science 97, 467 (1943); King, J. Chem. Soc. 1948, 1945; Bick, Clezy, ibid. 1960, 2402; Boissier et al., Lloydia 28, 191 (1965), C.A. 64, 948b (1966). Structure: Dutcher, J. Am. Chem. Soc. 68, 419 (1946); Hultin, Acta Chem. Scand. 15, 1130 (1961). Configuration: Bick, Clezy, J. Chem. Soc. 1953, 3893; Hultin, Acta Chem. Scand. 17, 753 (1963). Revised structure: Everett et al., Chem. Commun. 1970, 1020. 13C-NMR: L. Koike et al., J. Org. Chem. 46, 2385 (1981).
Properties
Slender needles from methanol, mp 232-234°; also reported as mp 218-220° (Boissier). [α]D24 +200° (c = 0.5 in 0.1N HCl). [α]D24 +105° (c = 0.9 in pyridine).Derivative
Sulfate tetrahydrate.C36H38N2O6.H2SO4.4H2O; mol wt 764.84.
C 56.53%, H 6.33%, N 3.66%, O 29.29%, S 4.19%.
Properties
Rectangular plates from water. Contains 9.43% H2O. After drying, mp 263-265° (dec). [α]D24 +184° (c = 0.375 in methanol).Derivative
Methiodide.Properties
mp 270-275°.Derivative
Dimethiodide.C38H44I2N2O6; mol wt 878.57.
C 51.95%, H 5.05%, I 28.89%, N 3.19%, O 10.93%.