2237. Chromocarb
Nomenclature
Description and references
Prepn: S. Ruhemann, H. E. Stapleton, J. Chem. Soc. 77, 1179 (1900); J. Schmutz et al., Helv. Chim. Acta 34, 767 (1951); G. Pifferi et al., J. Heterocycl. Chem. 14, 1257 (1977). Prepn of diethylamine salt: P. A. Tronche, ZA 6807352; eidem, US 3816470 (1969, 1974 both to Ferlux). Pharmacology of diethylamine salt in animals: J. Couquelet et al., C. R. Seances Soc. Biol. Ses Fil. 164, 329 (1970); P. Conquet et al., ibid. 800. Clinical comparison with dipyridamole of effect on platelet function: A. Vittoria et al., Curr. Ther. Res. 35, 1033 (1984). Clinical trial in diabetes with vascular disease: N. Ciavarella et al., ibid. 36, 293 (1984). Bioavailability in humans: J.-M. Aiache et al., Biopharm. Drug Dispos. 7, 301 (1986).
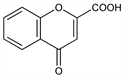
Properties
Colorless needles from alcohol, mp 250-251° (dec) (Ruhemann, Stapleton); also reported as mp 255-256° (Pifferi). uv max: 230, 305 nm (ε 20220, 8075). Sol in alcohol, ammonia. Sparingly sol in water.Derivative
Diethylamine.Nomenclature
Properties
Microcrystalline powder from alcohol + acetone, mp 138°. Sol in water. LD50 in mice: ≈800 mg/kg i.v.; >5 g/kg orally (Tronche, 1974).Therapeutic Category
Diethylamine salt as capillary protectant.
Keywords
Vasoprotectant